QIKcap System™
FDA De Novo Granted — The First and Only UV-C IV Port Microbial Reduction Device
The 10-second defense for every IV port
QIKcap™ is the first and only FDA-cleared UV-C device for luer-activated valves — eliminating compliance variability in central line maintenance and delivering verifiable, automated microbial reduction at point of care.
The Problem
Manual disinfection alone isn't enough.
Bloodstream infections (BSIs) remain one of the most costly and preventable hospital-acquired infections, averaging $48,000+ per bloodstream infection per the CDC. Despite "scrub the hub" protocols, compliance rates and technique vary dramatically — creating uncontrolled risk at every IV access event.
Human factors — friction, fatigue, urgency — make manual wipe protocols and alcohol port protectors an inconsistent last line of defense.
The Solution
Built for the bedside. Designed for compliance.
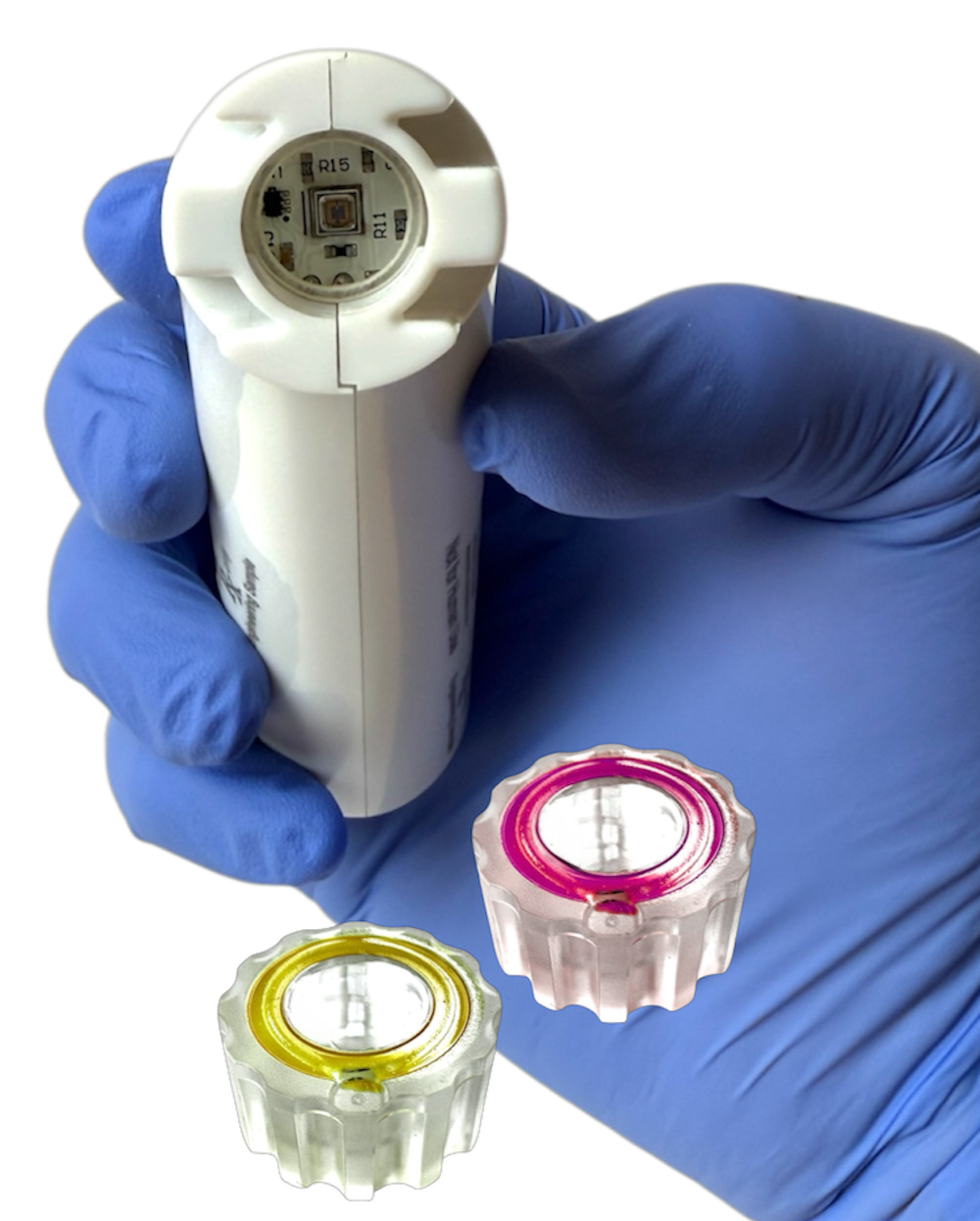
Automated UVC LED Technology
Rapid 10-second microbial reduction
Passive Protection Up to 7 Days
Ergonomic handheld design
Visual & Tactile cycle completion confirmation
Sterile singe-use caps
Universal intravenous luer cap
ergonomic IV pole strip wiht easy-peel pouch; strips of 7 caps
The Data
We didnt avoid the worst case. We tested it!
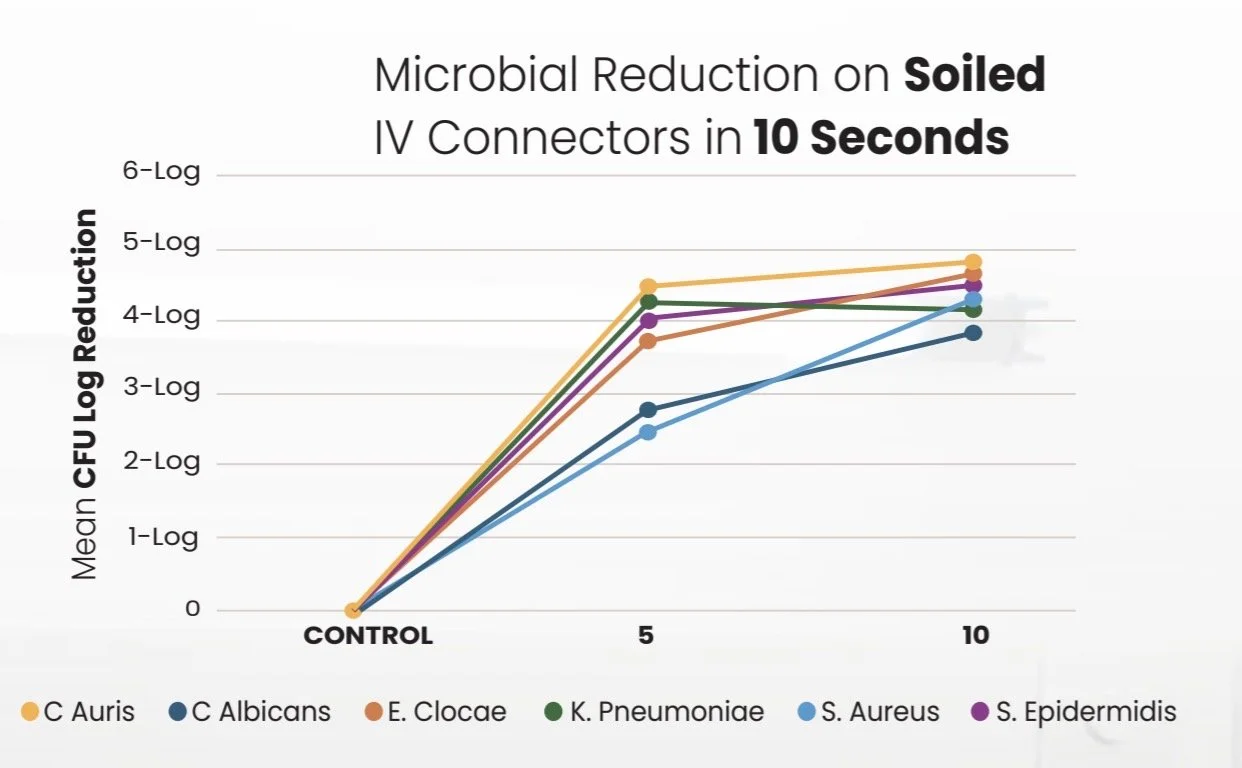
The QIKcap System was independently evaluated in two rigorous in-vitro studies to assess its antimicrobial efficacy and microbial reduction capability against clinically relevant pathogens on needleless connectors — a well-documented source of central line-associated bloodstream infections (CLABSIs).
We tested against extreme soiled conditions per quantitative suspension methodology with organic soil load simulating worst-case clinical contamination — conditions under which no IPA protective cap on the market has published comparable data.
Cap it & Zap it
Watch it Work
The Impact
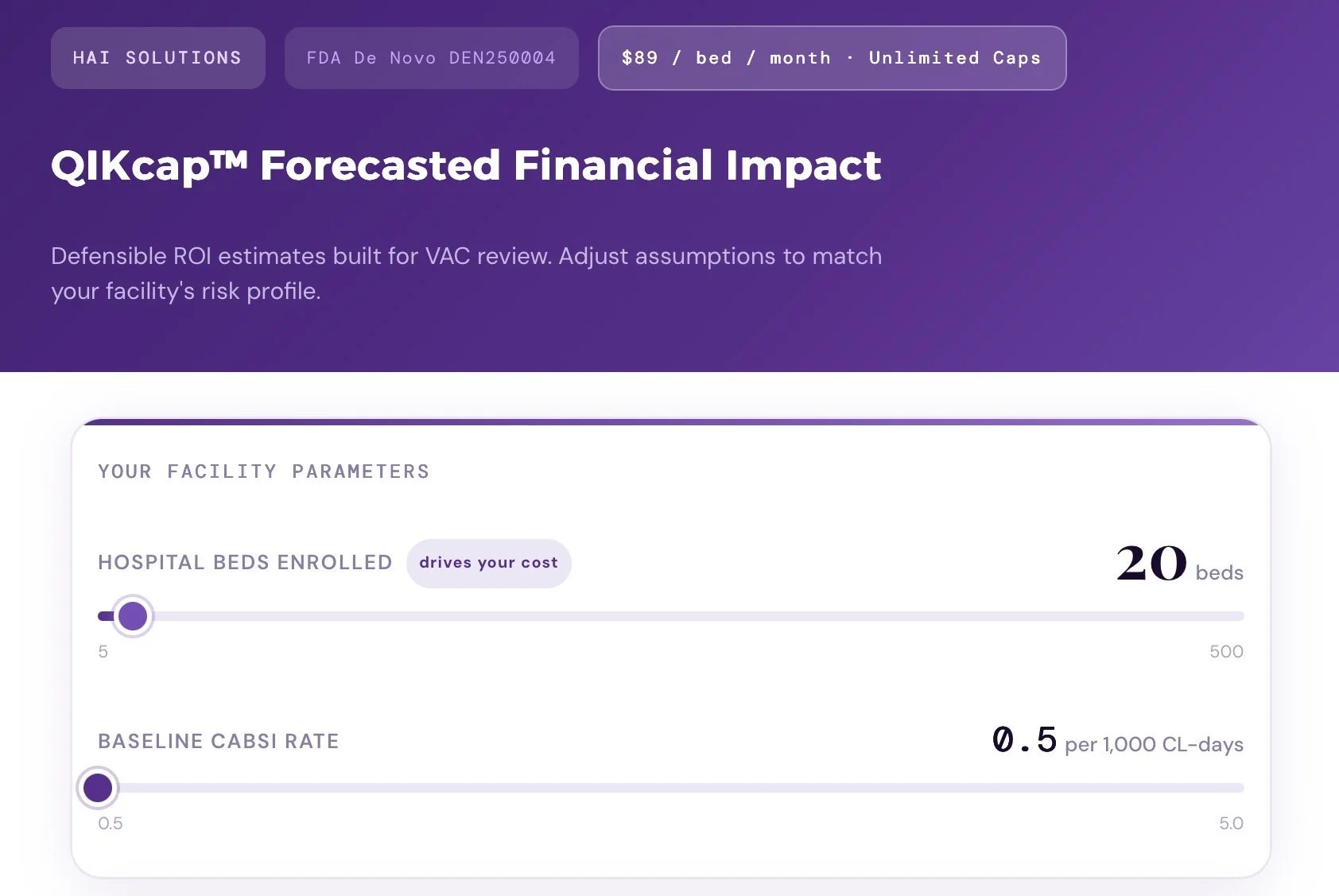
QIKulator is an interactive online ROI calculator designed to help hospitals estimate the financial impact of adopting QIKcap™.
The tool generates defensible ROI estimates specifically built to withstand scrutiny from hospital Value Analysis Committees (VAC). It focuses on reducing catheter-associated bloodstream infections (CABSI) by targeting hub/connector-related infections only, using conservative, literature-backed assumptions that users can customize to match their facility’s risk profile.
-
The QIKCAP Device, used in conjunction with the single-use QIKCAP Cap, is a UVC treatment device intended to supplement manual needle-free luer connector instructions to 'scrub the hub' with CHG/IPA wipes. After conducting the protocol in the needle-free connector instructions for use, the QIKCAP Device applies UVC to the connector septum for 10 seconds to assist in reducing microbial contamination. The single use QIKCAP Cap also functions as a physical barrier, helping protect the connector from environmental contamination for up to 7 days if not removed.
Under the conditions of testing, the combined use of an 3.15% CHG/70% IPA wipes applied for a 5 seconds and the QIKCAP Device resulted in 4-log reductions in Staphylococcus aureus, Klebsiella pneumoniae, Staphylococcus epidermidis, and Enterobacter cloacae. The device has not shown a significant reduction in fungal organisms. A correlation to clinical infection-related outcomes has not been established.
-
The reusbale DEVICE will vibrate and the an indicator light will turn green. Additonally the disposable CAP also includes a chromatic dosimeter which changes color from Yellow to Magenta confirming UVC cycle completion.
-
Yes, after following the IFU the IV connector can be immediately accessed.
-
Yes. Please review our QIKcap Forecasted Financial Impact QIKculator which allows you to set your bed size and facility metrics to assess total financial impact. We also support a sustainable cap model if your facility wishes to take advantage.
We are engineering patient safety
Schedule a trial evaluation, request clinical materials, or connect with our team. We work directly with infection prevention, nursing leadership, and value analysis committees.